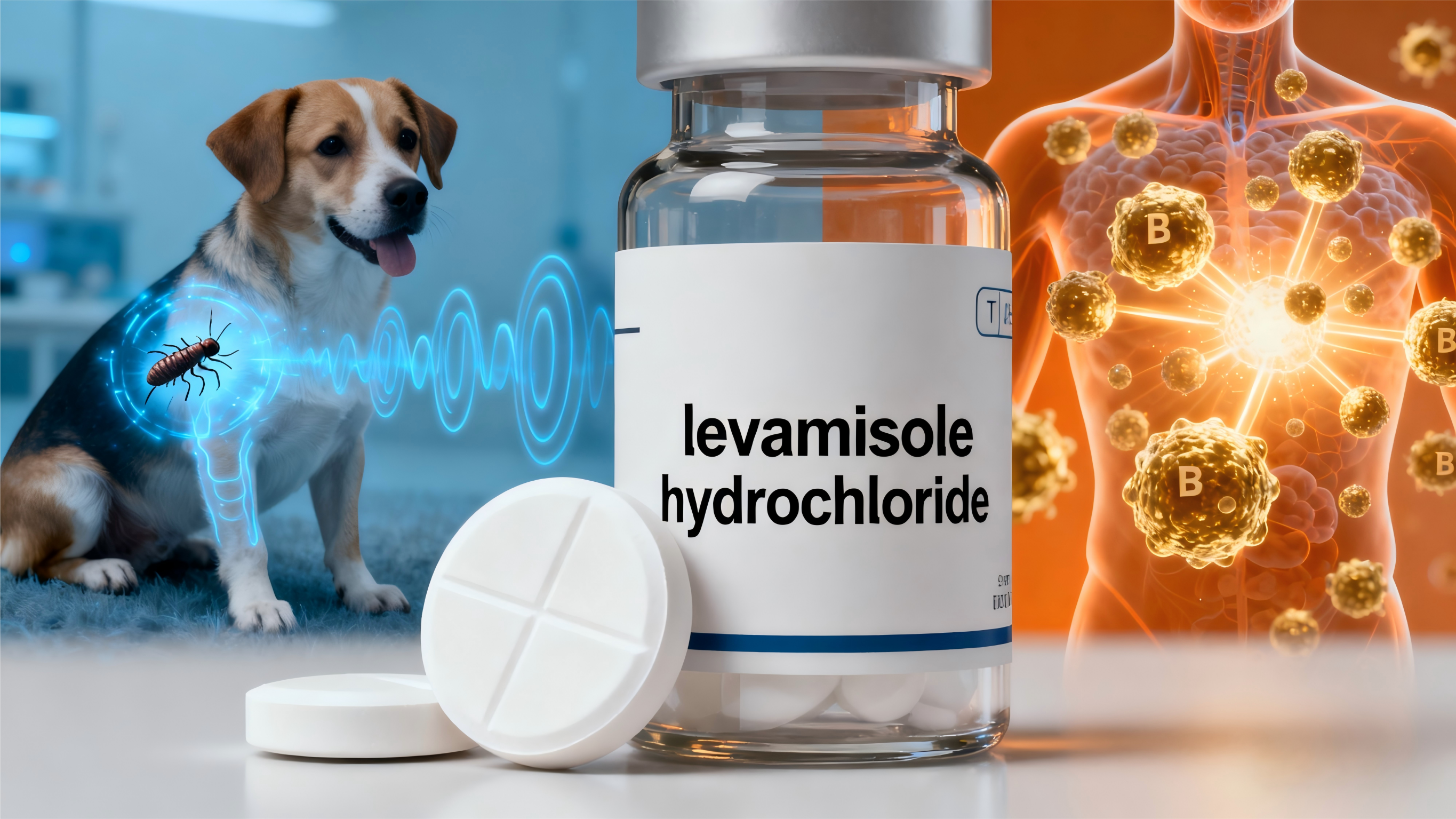
levamisole hydrochloride livestock
Levamisole hydrochloride livestock represents a cornerstone pharmaceutical solution in modern veterinary medicine, serving as a highly effective broad-spectrum anthelmintic agent specifically formulated for animal health applications. This pharmaceutical compound demonstrates exceptional efficacy against various parasitic infections that commonly affect domestic animals, including cattle, sheep, goats, swine, and poultry. The active ingredient, levamisole hydrochloride, functions through a unique mechanism of action that targets the nervous system of parasitic worms, causing paralysis and subsequent elimination from the host animal. This veterinary pharmaceutical has established itself as an indispensable tool for livestock producers seeking comprehensive parasite control solutions. The formulation exhibits remarkable versatility in its application methods, available in multiple administration forms including oral solutions, injectable formulations, and feed additives, allowing veterinarians and livestock managers to select the most appropriate delivery method based on specific animal requirements and operational preferences. Levamisole hydrochloride livestock demonstrates superior bioavailability characteristics, ensuring rapid absorption and distribution throughout the animal's system for maximum therapeutic impact. The compound's pharmacokinetic profile facilitates efficient elimination of target parasites while maintaining excellent safety margins when used according to established veterinary guidelines. Modern manufacturing processes ensure consistent quality and potency in every batch of levamisole hydrochloride livestock, with rigorous quality control measures implemented throughout production to guarantee pharmaceutical-grade standards. The product's stability profile allows for extended shelf life under proper storage conditions, providing livestock producers with reliable inventory management capabilities. Additionally, levamisole hydrochloride livestock incorporates advanced formulation technologies that enhance palatability and ease of administration, reducing stress on animals during treatment procedures while ensuring optimal therapeutic outcomes for comprehensive parasite management programs.