Olmesartan Medoxomil
API Supplier | High-Efficacy Angiotensin II Receptor Blocker (ARB API)
GTIG Guomian is your reliable Olmesartan Medoxomil API supplier and strategic partner. We specialize in supplying high-purity Angiotensin II Receptor Blocker (ARB) API compliant with USP/EP standards. Sourced from certified GMP manufacturers and backed by complete registration documentation (including open Olmesartan Medoxomil DMF/EDMF files), we ensure a stable supply to global markets such as Latin America, Asia, the Middle East, and Africa. [Looking for a compliant, competitive source of Olmesartan Medoxomil API? Request a quote or apply for a free sample now!
- Overview
- Recommended Products
- 💊Prescription Drug Formulations: As the Active Pharmaceutical Ingredient (API), specifically used in producing single-component or combination preparations (e.g., combined with amlodipine) for the treatment of essential hypertension, primarily in tablet form.
- 🌍Global Market Access: Our product complies with multiple pharmacopoeial standards and comes with complete registration document support, designed to help your finished product smoothly enter emerging markets such as Latin America (e.g., Brazil), Asia (e.g., India, Southeast Asia), the Middle East, and Africa.
- 🏭Stable Supply, Production Assurance: Integrate resources from high-quality cGMP manufacturers to ensure long-term, stable supply of antihypertensive API.
- 📑Compliance Support, Faster Time-to-Market: Provide complete registration documentation (including open Olmesartan Medoxomil DMF files) to facilitate rapid international registration with authorities like Brazil's ANVISA and India's CDSCO.
- ✈️Global Logistics, Optimized Capital: Leverage an efficient global network and flexible inventory planning, supporting both small sample batches and large commercial orders.
- 🎯Take Action Now: [Contact Us for the Latest Olmesartan Medoxomil Quotation] or [Apply for a Free Olmesartan Medoxomil Sample (with COA)]
- Quality Assurance: Stringent control under our cGMP quality system ensures high consistency and traceability of product quality for every batch.
- Stable Production Capacity: We have reliable manufacturing partners and a robust supply chain, effectively guaranteeing a long-term, stable supply of this critical API (annual capacity exceeding 30 tons).
- Specification Flexibility: We offer various specifications from 100g R&D grade and 1kg samples to 25kg commercial packaging, meeting your needs from R&D to commercial production stages.
- Comprehensive Technical Documentation: Provide detailed Olmesartan Medoxomil COA and MSDS (Material Safety Data Sheet).
- Registration Document Support: Provide relevant open sections of DMF/EDMF files compliant with international markets, professionally supporting registrations with authorities like Brazil's ANVISA and India's CDSCO.
- Regulatory Consultation: Our regulatory affairs team can provide preliminary consultation on import regulations for target markets (e.g., Brazil ANVISA, India CDSCO).
- Efficient Global Network: Partner with professional chemical logistics providers to ensure full control from factory to customer.
- Key Market Expertise: Proficient in customs clearance procedures for shipments to Brazil, India, Southeast Asia, the Middle East, and Africa.
- Reliable Delivery Commitment: Provide clear delivery timelines and real-time cargo tracking.
- Q: Can you provide Olmesartan Medoxomil samples for quality verification?
A: Yes. We are happy to provide free samples (typically 1 kg) to potential customers, accompanied by the corresponding batch COA for testing. - Q: What is the delivery lead time for Olmesartan Medoxomil API?
A: Commercial orders are typically shipped within 3-4 weeks after order confirmation, depending on order volume and destination customs requirements. - Q: What quality documents are provided upon purchase?
A:COA and MSDS are provided with shipment. DMF/EDMF files for registration can be provided as needed (open sections). - Q: Do you support international registrations like Brazil ANVISA and India CDSCO?
A: Yes, we can provide a technical document package (including open sections of DMF/EDMF) that meets the requirements of Brazil's ANVISA and India's CDSCO, supporting your product's international registration.
1. Product Summary
|
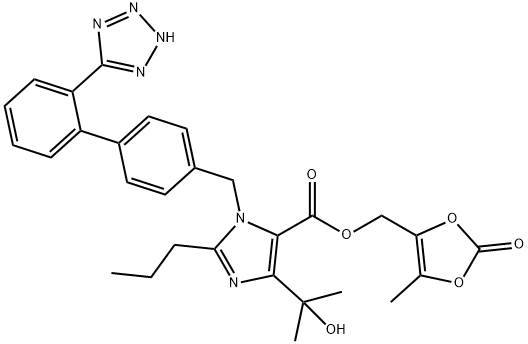
Product Name: Olmesartan Medoxomil CAS No.: 144689-63-4 Molecular Formula: C29H30N6O6 Molecular Weight: 558.59 |
 |
Grade/Standard: Pharmaceutical Grade, compliant with USP/EP standards
Appearance: White or almost white crystalline powder
Minimum Order Quantity (MOQ): 1 kg (sample), 25 kg (commercial order, LCL service supported)
2. Product Overview
Olmesartan Medoxomil is an important prodrug that is rapidly hydrolyzed in vivo to its active metabolite, olmesartan, after oral administration. Olmesartan is a specific Angiotensin II Receptor Blocker (ARB) that effectively dilates blood vessels and lowers blood pressure by selectively blocking the binding of angiotensin II to the AT1 receptor. As a modern first-line antihypertensive drug, it is characterized by significant efficacy and good tolerability.
As a professional pharmaceutical API supplier, we deeply integrate resources from top-tier domestic cGMP Olmesartan Medoxomil manufacturers. We provide stable, compliant USP/EP-grade Olmesartan Medoxomil to global antihypertensive drug manufacturers, making us your trustworthy partner for antihypertensive API.
3. Key Application Areas
4. Key Value Proposition Summary
5. Three-in-One Solution Package
a. Manufacturer Resource Integration Advantage
b. Professional Compliance & Registration Support
c. Supply Chain & Logistics Services
6. Packaging & Shipping
We use moisture-proof, light-resistant, vacuum-sealed or nitrogen-purged pharmaceutical-grade packaging to ensure the chemical stability of the product during transport and storage. Standard commercial packaging is 25 kg/carton drum (with inner polyethylene bag). R&D and sample packaging options include 100 g/bottle or 1 kg/aluminum foil bag.
7. Frequently Asked Questions (FAQ)
Looking for a strategic partner in the cardiovascular disease treatment field? GTIG Guomian is your ideal Olmesartan Medoxomil supply chain partner.
[Contact Us Now for Customized Solutions and a Free Sample!]
Olmesartan Medoxomil API Technical Parameters (EP)
Items |
Specification |
Results |
Appearance |
White or almost white crystalline powder |
Visual Inspection |
Solubility |
Practically insoluble in water, slightly soluble in ethanol (96 per cent), practically insoluble in heptane. |
EP 5.11 |
Identification |
IR: Infrared absorption spectrum of the substance corresponds to that of olmesartan medoxomil CRS. |
EP 2.2.24 |
Related compounds |
Impurity A: Not more than 0.4% |
EP 2.2.29 |
Impurity C: Not more than 0.3% | ||
Unspecified impuritites: Not more than 0.10% | ||
Total impurities: Not more than 0.7% | ||
Water |
Not more than 0.5% |
EP 2.5.32 |
Sulfated ash |
Not more than 0.1% |
EP 2.4.14 |
Assay |
97. 5%--102.0%, calculated on the anhydrous substance 100.1% C₂₉H₃₀N₆O₆ |
EP 2.2.29 |
Residual solvents |
Acetone: Not more than 6000ppm |
Internal Standard |
Tetrahydrofuran: Not more than 720ppm | ||
Ethyl Acetate: Not more than 5000ppm | ||
Methanol: Not more than 3000ppm | ||
Dichloromethane: Not more than 600ppm | ||
Ethanol: Not more than 5000ppm | ||
N,N-Dimethylformamide: Not more than 880ppm | ||
N-Nitroso- |
Not more than 0.03ppm |
NA |
N-Nitrosodiethyla |
Not more than 0.03ppm |
NA |
NDMA+NDEA |
Not more than 0.03ppm |
Japanese Market |
Particle Size Distribution |
Reference |
As Requested by Customer |
Triphenylmethyl Azide |
Not more than 37.5ppm |
Japanese Market |
Microbial |
Total aerobic microbial count NMT 1000CFU; |
Internal Standard |
Basis for Standard Establishment |
The in-house specification for Olmesartan Medoxomil was established in accordance with the European Pharmacopoeia monograph (01/2017:2600). |
|
Document generation date: January 7, 2026
© 2026 GTIG Guomian – Professional Pharmaceutical API and Specialty Chemical Supplier