Valsartan
High Purity Angiotensin II Receptor Blocker for Hypertension Treatment
GTIG Guomian is your reliable Valsartan API supplier and strategic partner. We specialize in supplying high-purity Angiotensin II Receptor Blocker APIs compliant with USP/EP pharmacopoeia standards, suitable for antihypertensive drug and cardiovascular treatment product development.
Our products originate from certified cGMP facilities, supported by complete Valsartan DMF documentation, ensuring stable supply to global markets.
- Overview
- Recommended Products
- Quality Assurance: Strict control under our cGMP quality system, with special attention to controlling nitrosamine impurities (e.g., NDMA), ensuring safety standards and quality consistency for each batch.
- Stable Production Capacity: We have reliable production partners and optimized supply chains, effectively ensuring long-term, stable supply of this critical cardiovascular API. (Annual production capacity exceeds 400 tons)
- Specification Flexibility: We offer multiple specifications from 100g R&D grade, 1kg samples to 25kg commercial packaging, meeting your different stage needs from R&D to commercial production.
- Complete Technical Documentation: Each batch of products comes with detailed Certificate of Analysis (COA) and Material Safety Data Sheet (MSDS), including nitrosamine impurity test reports.
- Registration Documentation Support: We can provide qualified partners with open parts of DMF/EDMF documentation required for drug registrations in the US, EU, and other international markets, significantly accelerating your product registration process.
- Regulatory Consultation: Our regulatory affairs team can provide consultation on the latest regulatory requirements for sartan drugs in target markets (e.g., Brazil ANVISA, India CDSCO).
- Partnership with professional chemical logistics companies covering key markets like Europe, Asia-Pacific, and Latin America.
- Proficient in customs clearance procedures, providing full cargo tracking and delivery commitments.
1. Core Product Summary
|
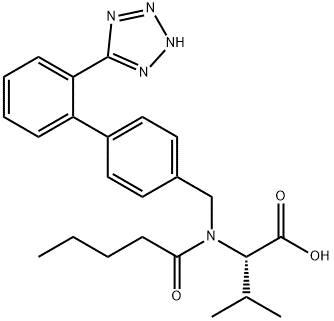
Product Name: Valsartan CAS No.: 137862-53-4 Molecular Formula: C₂₄H₂₉N₅O₃ Molecular Weight:435.52 |
 |
Grade/Standard: Pharmaceutical Grade, Compliant with USP/EP Standards
Appearance: White to off-white crystalline powder
Minimum Order Quantity: 100g (R&D), 1kg (Sample), 25kg (Commercial orders, LCL supported)
2. Product Overview
Valsartan is a widely used core Active Pharmaceutical Ingredient (API) in antihypertensive prescription drugs, belonging to the Angiotensin II Receptor Blocker (ARB) class. It effectively blocks angiotensin II, providing excellent blood pressure reduction and cardiorenal protection.
As your reliable Valsartan API supplier, we provide raw materials strictly compliant with USP/EP pharmacopoeia standards, supported by complete Valsartan DMF documentation, designed specifically for producing high-quality antihypertensive tablets, capsules, and combination formulations (such as Valsartan/Hydrochlorothiazide tablets), helping pharmaceutical companies meet stringent global market requirements for sartan drug impurity control.
3. Primary Application Areas
Prescription Antihypertensive Drugs: As a core antihypertensive drug ingredient, widely used in producing prescription tablets and capsules for treating essential hypertension.
Global Market Access Support: Products comply with multiple pharmacopoeial standards and provide complete registration documentation support (including DMF), facilitating market entry into Europe, Asia, Latin America, and other major global markets.
Combination Formulations and Combination Therapy: Suitable for developing combination formulations with components like hydrochlorothiazide and amlodipine, enhancing therapeutic efficacy and patient compliance.
4. Core Value Summary
Stable Supply, Ensuring Production: Relying on reliable production partners and supply chain systems, ensuring long-term stable supply of antihypertensive APIs, with annual production capacity exceeding 400 tons.
Compliance Support, Accelerating Market Entry: Providing complete Valsartan DMF documentation, supporting international registration submissions like EDQM, Brazil ANVISA, India CDSCO, helping shorten your product's time-to-market.
Global Logistics, Flexible Delivery: Efficient global logistics network, supporting fully controlled transportation from R&D samples to bulk orders.
5. Trifecta Solution
Raw Material Supply Advantages
Professional Compliance Support
Supply Chain and Logistics Service Solutions
6. Packaging and Transportation
We use moisture-proof, light-proof, vacuum or nitrogen-filled pharmaceutical-grade packaging, ensuring product chemical stability during transportation and storage, and minimizing impurity formation risks. Standard commercial packaging is 25kg/cardboard drum (with inner plastic bag liner), while R&D and samples are provided in 100g/bottle or 1kg/aluminum foil bag packaging.
7. Frequently Asked Questions (FAQ)
Q: Can you provide Valsartan samples for quality verification?
A: Yes. We provide free Valsartan samples (typically 100g) to potential customers, along with the corresponding batch's COA (Certificate of Analysis) for testing verification.
Q: What are the price and bulk purchase policy for Valsartan API?
A: We offer competitive Valsartan pricing, with better prices for bulk purchases. Specific MOQ (Minimum Order Quantity) and pricing please consult our sales team. We provide customized quotations based on your purchase volume and long-term cooperation intentions.
Q: What quality documents are provided upon purchase?
A: We provide COA (Certificate of Analysis) and MSDS (Material Safety Data Sheet) with shipments. Registration DMF/EDMF documentation can be provided upon request for open parts.
Q: What is the mechanism of action of Valsartan?
A: Valsartan is an Angiotensin II Receptor Blocker (ARB drug) that works by blocking the action of angiotensin II, lowering blood pressure, reducing cardiac workload, and achieving cardiovascular protection.
Q: What are the impurity control standards for Valsartan API?
A: We strictly follow USP/EP pharmacopoeial standards, with special attention to controlling nitrosamine impurities (e.g., NDMA), ensuring product safety and compliance.
8. Valsartan API Technical Parameters (USP/EP Specifications)
Items |
Specification |
Remarks |
Description |
White or almost white hygroscopic powder |
USP |
Solubility |
Freely soluble in anhydrous ethanol; sparingly soluble in methylene chloride; practically insoluble in water |
USP |
Identification |
A.Infrared Absorption spectrum is identical to that of Valsartan reference standard |
USP |
B.The retention time of the major peak of the sample solution corresponds to that of the standard solution, as obtained in the Assay |
USP |
|
Absorbance |
Not more than 0.02 at 420nm (1/20 methanol solution) |
USP |
Water |
Not more than 2.0% |
USP |
Azide |
Not more than 4.7ppm |
Internal Standard |
Related compounds |
Related compound A: Not more than 1.0% |
USP |
Related compound B: Not more than 0.2% | ||
Related compound C: Not more than 0.1 % | ||
Any other individual impurity: Not more than 0.10% | ||
Total impurities (excluding related compound A): Not more than 0.3% | ||
Residual solvents |
Methanol: Not more than 3000ppm |
Internal Standard |
Ethyl acetate: Not more than 5000ppm | ||
Methylene chloride: Not more than 600ppm | ||
Toluene: Not more than 890ppm | ||
N,N-Dimethylformamide: Not more than 880ppm | ||
Residue on Ignition |
Not more than 0.1 % |
USP |
Particle Size Distribution |
Reference |
As Requested by Customer |
Bulk Density |
Reference |
As Requested by Customer |
Tapped Density |
Reference |
As Requested by Customer |
Assay |
98.0%---102.0% of C₂₄H₂₉N₅O₃ calculated on the anhydrous basis |
USP |
N-Nitroso- |
Internal Standard |
|
N-Nitrosodiethyla |
Internal Standard |
|
Azidosartan Biphenyl |
Not more than 4.7ppm |
Internal Standard |
Azidosartan Biphenyl Tetrazolium |
Not more than 4.7ppm |
Internal Standard |
Crystal Form |
The peak positions in the XRPD pattern of this product correspond to those reported for the predominantly amorphous form in Patent US7105557B2. |
As Requested by Customer |
Microbial Limit |
Not more than 10 cfu of total aerobic microbial count in 1 g of the sample; not more than 10² cfu of total combined yeasts and moulds count in 1 g of the sample; Escherichia coliabsent in 1 g of the sample. |
* |
Standard Established |
The internal standard was established in accordance with the USP monograph for Valsartan |
|
9. Contact Us
Looking for a strategic partner in cardiovascular treatment drugs?
GTIG Guomian is your ideal Valsartan API supply chain partner.
Contact us now for customized solutions and free samples!
Document generation date: January 7, 2026
© 2026 GTIG Guomian – Professional Pharmaceutical API and Specialty Chemical Supplier